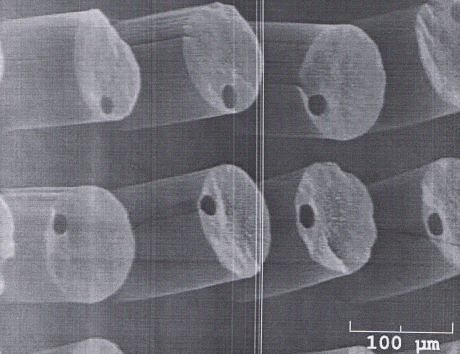
Jud Ready from the Georgia Tech Research Institute has developed a carbon nanotube (CNT) array to provide electrical stimulation and read-out electrodes for drug-eluting nano-channel neuronal interfaces. A densified array of CNT towers form spikes for use as electrically conductive neural probes. A lithographic pattern technique is used to create toroidal syringes and inductively coupled plasma is used to micromachine the underlying Si substrate with fluid delivery channels. This results in CNT arrays composed of densely packed CNT “vines” which allow weepage of the drug from the sidewalls of the array for better bio-incorporation.
- Densely packed CNT arrays allow for better bio-incorporation, prevention of glial scarring, chronic inflammation, and neuronal cell loss
- Readily functionalized to promote neuronal growth and tissue-electrode adhesion, enhancing electrode performance and quality of read-out signals
- Inherent mechanical stiffness enhances the rigidity of adjacent CNTs preventing buckling as in other high-aspect probe systems.
- Promote neuronal growth and adhesion between the electrode and biologic tissues
- “Nanosyringes” for drug elution
Microelectrode arrays (MEAs) are neural interfaces that connect neurons to electronic circuitry and include implantable interfaces such as deep brain stimulators, cochlear implants, and cardiac pacemakers. Research suggests that MEAs provide insight into memory function and perception, and therapeutic value for conditions such as epilepsy, depression, and obsessive compulsive disorder (OCD). MEAs can record activity from multiple neurons simultaneously, but electrode implantation can cause chronic inflammation, glial scarring, neuronal cell loss, and a decrease in axonal growth. Current metallic electrode arrays are too large in diameter for applications requiring biocompatibility, which may result in glial scarring at the electrode penetration site leading to decreased performance over time and complete neural degeneration. Attempts to create smaller electrode arrays have failed due to the high aspect ratio that leads to buckling during insertion through various tissue layers.