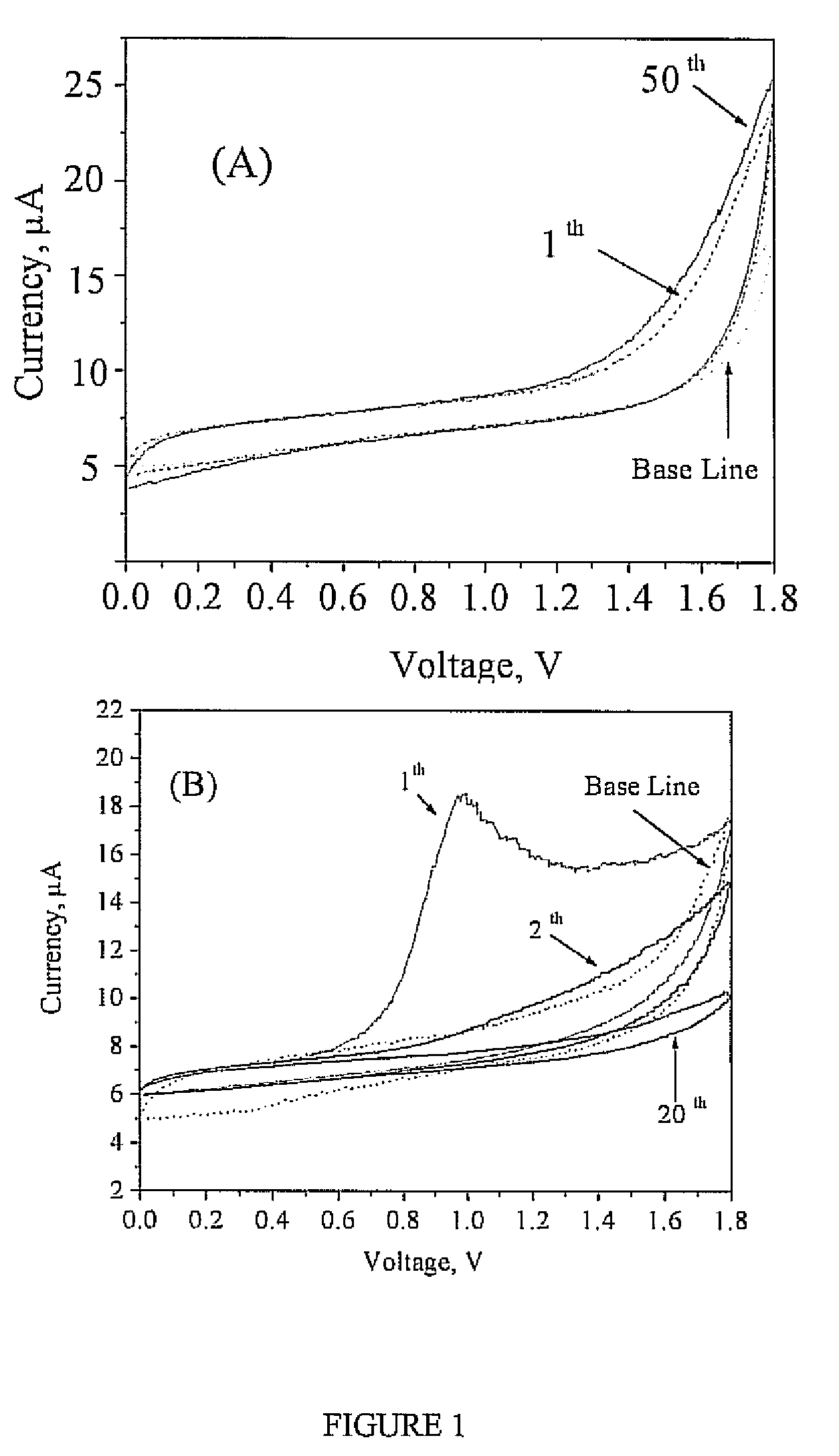
Georgia Tech inventors have developed a new class of hybrid inorganic-organic proton conducting polymers containing a backbone with an attached heterocyclic compound with sulfonyl functionality. These polymers offer high proton conductivity and thermal stability up to 250 °C in low-humidity environments and good mechanical properties and water resistance. Proton conductivities increase with phosphoric acid (H3PO4) content and temperature, reaching 3.2×10−3 S/cm at 110 °C in a dry atmosphere for a membrane with 1 mole of imidazole ring and 7 moles of H3PO4. The proton conductivity increases with relative humidity (RH) as well, reaching 4.3×10−2 S/cm at 110 °C when the RH is increased to about 20%. They are ideally suited for use as electrolytes in high-temperature PEM fuel cells.
- High proton conductivity
- Thermal stability
- Strong mechanical properties
- Water resistant
- Fuel cells
- Purification and reforming cells
- Electrochemical devices
Various conductive polymers and metals are typically used in PEMs; however, conventional materials all have some disadvantages. Some polymers are not electrochemically stable, while precious metal membranes may experience oxidation or a loss of efficiency, particularly at high temperatures or at high humidity. This technology enables the development of polymers with stable conductive properties comparable to those of metallic membranes, making them well suited to the fuel cell environment.